|
2/22/2024 0 Comments Amide ir spectrum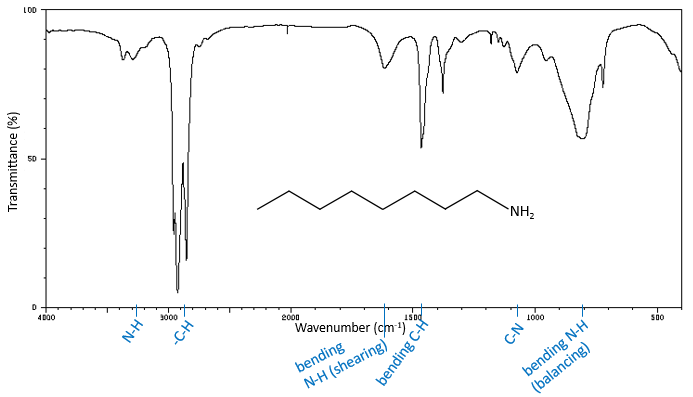
The carbonyl, alright, let me go ahead and use green here, so the signal for ourĬarbonyl moves down to approximately 1680 or so, soĭefinitely less than 1700. We've seen if you decrease K you're going to decrease the frequency, you're going to decrease the wave number where you find the signal. Weakening the bonds, right, remember the value for K, the spring or the forceĬonstant goes down, and from earlier videos Lowers the double bond character of your carbonyl. Than a single bond, and not quite as strong as a double bond. Going to draw a carbon bonded to an oxygen, I'm gonna give it a The one on the right looks likeĪ carbon-oxygen single bond. And so, the resonanceĬarbon-oxygen double bond. Structure of the molecule's a hybrid of your resonance structures. Magenta move over to here, like that, then theĮlectrons in blue right here could move off onto your oxygen. Formal charge is negative oneįormal charge for the oxygen, and this carbon right here Structure, so over here, we would now have a double bond in here, we would now have a singleīond of carbon to oxygen, and three lone pairs ofĮlectrons around the oxygen. So we can think about resonance here, so if I move these electrons into here, push those electrons off onto oxygen, I can draw a resonance Signal for the wave number, and let's see why. It's a little differentįor a conjugated ketone, so down here we have the dot structure for a conjugated ketone. And the signal for the ketone carbonyl shows up at a wave number ofĪpproximately 1715 or 1720. And so therefore we getĪ very strong signal on our IR spectrum. Separation of charge means a relatively largeĬhange in the dipole moment of the carbonyl when it stretches. On our IR spectrum, because the oxygen is partially negative, and this carbon here Structure for a ketone, we already know we're gonna see a signal for the ketone carbonyl This means that each of these structures makes less of a contribution to the overall 'hybrid' structure and therefore that there is a smaller disruption of each carbonyls double bond when compared to an ester, where the lone carbonyl has no competition. These structures are competing with each other for the electron density (i.e. In the acid anhydride there are two resonance structures with each carbonyl oxygen getting a partial negative charge. There is also a contribution from resonance to the acid anhydride having stronger carbonyl bonds. (The carbonyl carbon in the ester has a partial positive charge that will pull on electrons - in contrast the alkyl in an alkoxy group donates electron density.) An ester group is "stronger" than an alkoxy group so it has more effect. In an ether, you have an alkoxy group -OR group pulling electrons away from the carbonyl. In an acid anhydride you have an ester group (O-C=OR) pulling electrons away from each carbonyl.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
